Before going into details, it is important to understand the big picture in very simple terms. Both uranium and thorium are used to produce nuclear energy, but they behave differently inside a reactor. Uranium (especially U-235 or plutonium) can directly undergo fission, meaning it can split on its own in a reactor and release energy. Thorium, on the other hand, cannot do this directly. It must first be converted into another material—Uranium-233 (U-233)—through a process called breeding. Only after this conversion can energy be produced.
This difference leads to important consequences. The process becomes more controlled in thorium systems because fuel is created step-by-step inside the reactor rather than being fully ready from the start. This affects safety, weaponization potential, and nuclear waste. Uranium systems are simpler but produce more long-lived waste and can be adapted more easily for weapons. Thorium systems are more complex but tend to be harder to misuse and cleaner in waste profile.
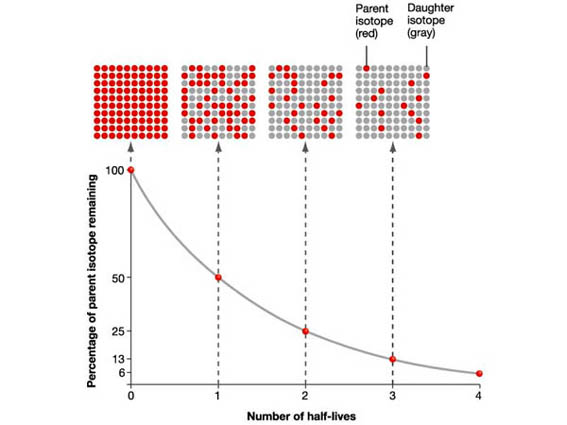
⏳ How Half-Life Connects to Element Conversion
4
Half-life is the time required for half of a radioactive substance to transform into another substance. This is not just a theory—it directly controls how thorium becomes usable fuel inside real reactors.
In a thorium system, after absorbing a neutron, Thorium-232 becomes Thorium-233, which has a half-life of about 22 minutes. This quickly decays into Protactinium-233, which has a much longer half-life of about 27 days. Only after this delay does it finally become Uranium-233, the actual fuel.
This time gap is very important in real reactor design. For example, in advanced thorium reactor concepts like India’s AHWR (Advanced Heavy Water Reactor), engineers must carefully manage protactinium because if it absorbs another neutron before decaying, it will not form useful fuel. Some reactor designs even consider isolating protactinium temporarily to allow it to decay safely into U-233.
This shows how half-life is not just a property—it is a design parameter that determines how efficiently fuel is produced and how stable the reactor remains.
🔬 How Uranium Produces Energy (Direct Fission)
4
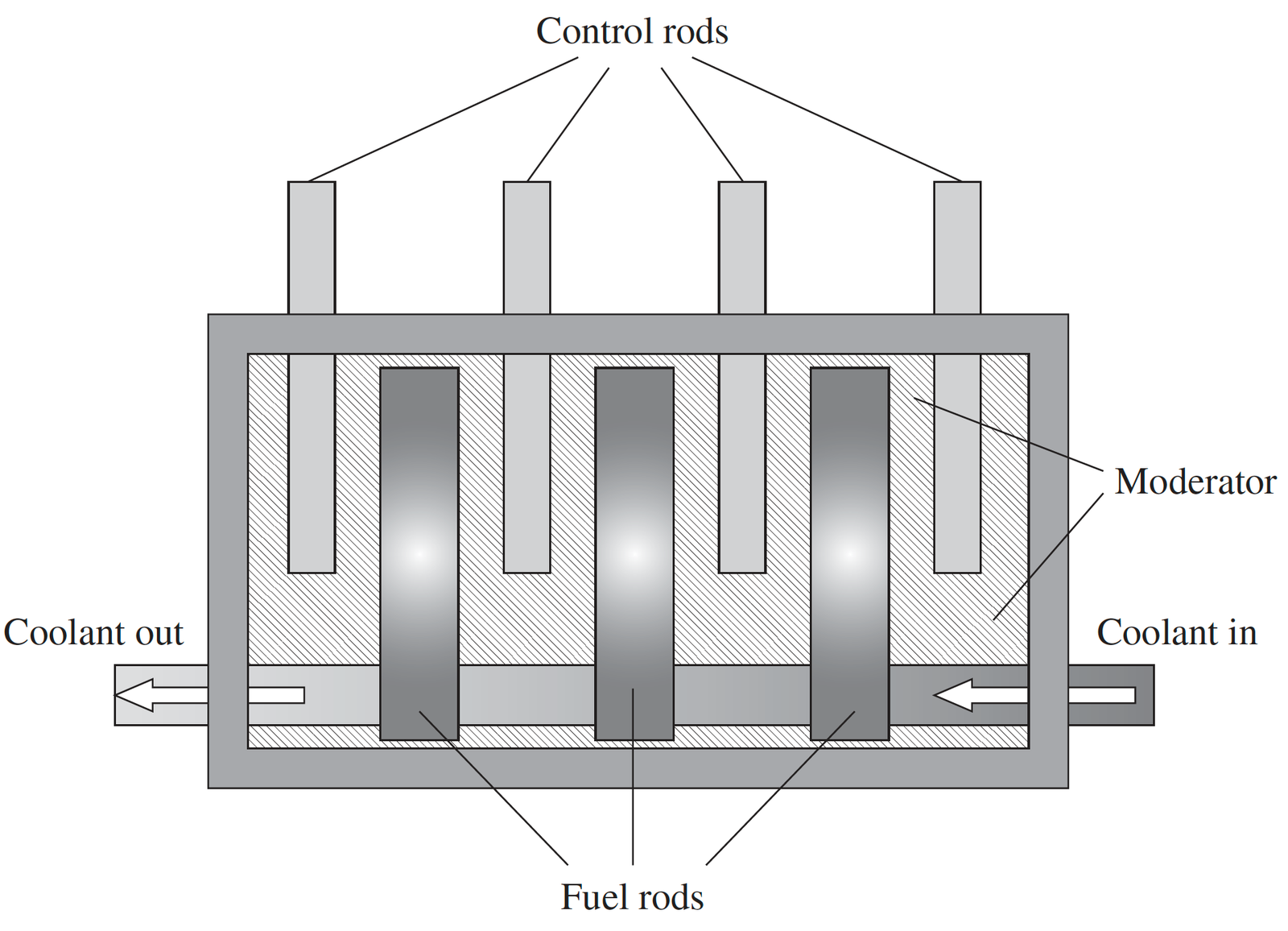
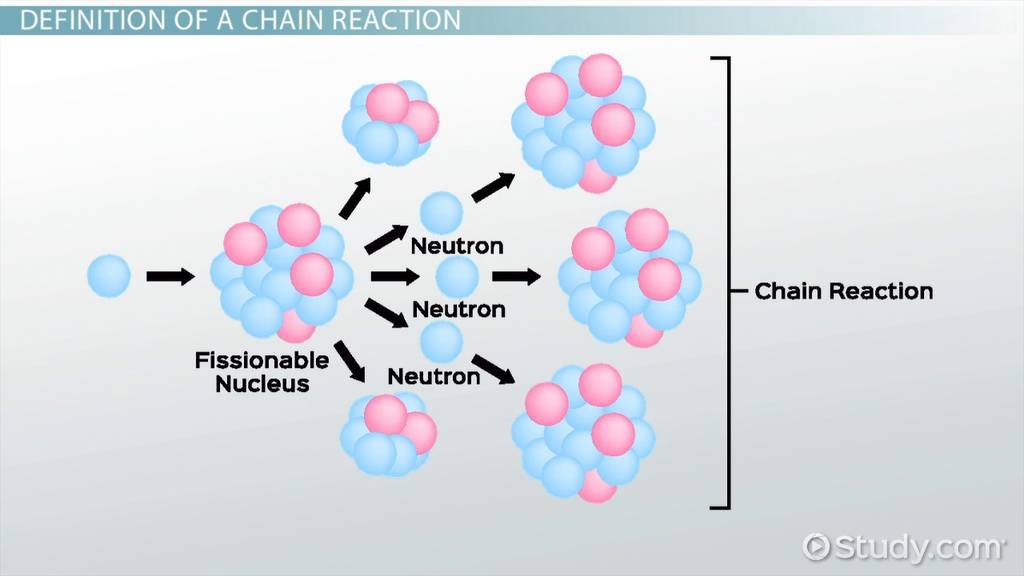
In a uranium-based reactor, the process is relatively direct. Certain types of uranium, especially U-235, are naturally capable of splitting when hit by a neutron. When one atom splits, it releases energy and additional neutrons, which go on to split more atoms. This creates a self-sustaining chain reaction.
Because the fuel is already capable of fission, the reactor mainly focuses on controlling the reaction using control rods and cooling systems. However, this also means that if control is lost, the reaction can escalate quickly, which is why uranium reactors require very strict safety systems.
During reactor operation, some uranium atoms also absorb neutrons without splitting and gradually transform into plutonium-239, which becomes a critical by-product.
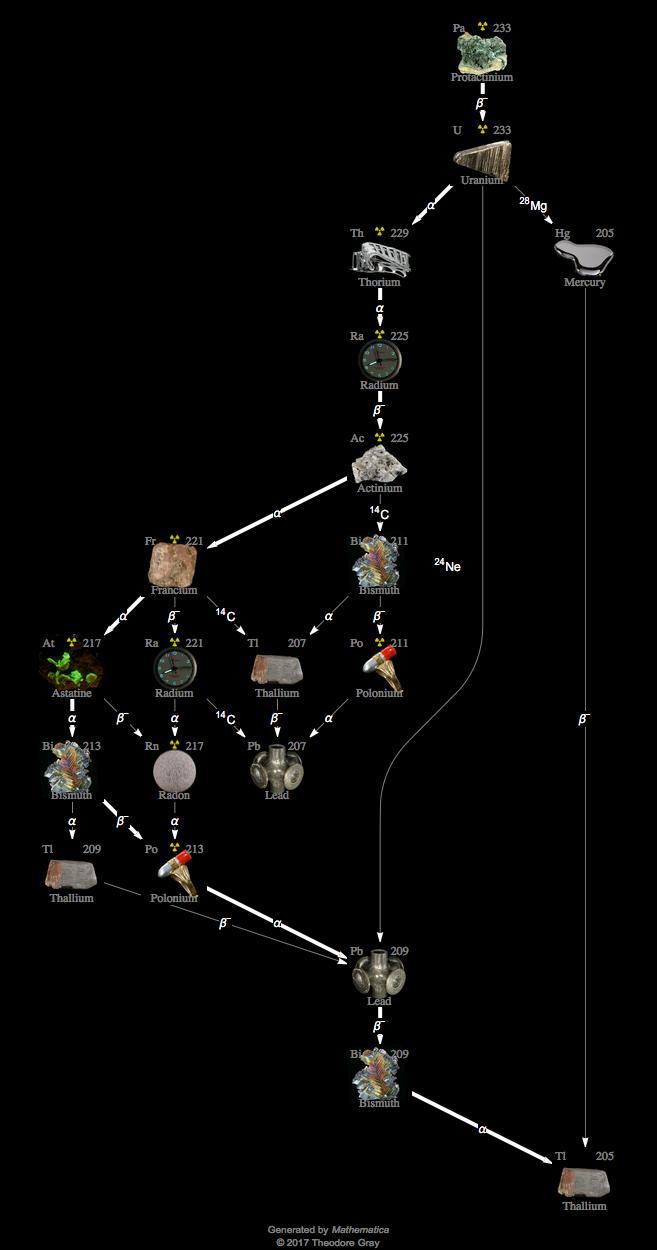
🔁 What Thorium Does Differently (The Breeding Process)

4
Thorium changes the process fundamentally. Thorium-232 cannot undergo fission directly, so it must first be transformed. This happens when it absorbs a neutron and slowly changes into Uranium-233, which is capable of fission.
This transformation happens in stages: thorium becomes thorium-233, then protactinium-233, and finally uranium-233. This entire sequence is called breeding, because the reactor is essentially creating its own fuel from a non-fissile material.
However, this process is technically complex. The reactor must maintain a precise neutron balance, because too few neutrons slow down fuel production, while too many can lead to unwanted reactions. Managing intermediate materials like protactinium is also challenging, as it can absorb neutrons and reduce efficiency. Fuel handling, reprocessing, and reactor design must all work together very precisely.
India’s progress in this area is considered significant because it has successfully developed fast breeder reactors and thorium-compatible designs, mastering a process that only a few countries have approached at scale. This is why breeding technology is seen as a major scientific and engineering achievement.
⚖️ Process Advantage: Why Use Thorium If It Becomes Uranium Anyway?
4
At first glance, it may seem that thorium offers no advantage since it ultimately becomes uranium-233, which then undergoes fission. However, the key difference lies in how and when the fuel is available.
In uranium reactors, fissile material is already present in significant amounts from the beginning. In thorium systems, the fissile material (U-233) is produced slowly and continuously. This creates a more stable and controllable reaction environment, because the fuel supply is linked to reactor conditions rather than being fully loaded at once.
This staged production acts like a natural regulator. Because fuel creation depends on neutron flow and decay timing, the system is less prone to sudden runaway reactions.
☢️ Weaponization: What Materials Are Produced and How They Differ
4
Different reactor types produce different by-products, and some of these materials can be used in nuclear weapons under specific conditions.
In uranium-based reactors, plutonium-239 is produced when uranium-238 absorbs neutrons. If fuel is removed early (a process called low burnup), the plutonium contains a higher proportion of Pu-239, which is suitable for weapons. Typically, a few kilograms of high-purity Pu-239 can be sufficient for a nuclear weapon, which is why such reactors are closely monitored internationally. Uranium itself can also be used in weapons, but only after enrichment to very high levels (above 90% U-235).
In thorium-based systems, the main fissile material produced is Uranium-233. In principle, U-233 can be used in weapons, and historically it has been tested. However, in practical reactor conditions, it is almost always mixed with Uranium-232, which emits strong gamma radiation. This radiation makes the material extremely difficult to handle safely and very easy to detect, which acts as a strong deterrent.
Additionally, the thorium fuel cycle does not naturally produce significant amounts of plutonium, removing one of the primary pathways used in traditional nuclear weapons programs. While no nuclear material is completely “weapon-proof,” thorium cycles are considered much less suitable for weaponization in real-world conditions.
♻️ Waste Management: By-products and Their Effects

4
All nuclear systems produce radioactive by-products, but their type, quantity, and lifetime differ significantly.
In uranium-based reactors, major by-products include plutonium-239, cesium-137, strontium-90, and minor actinides such as americium and neptunium. Plutonium remains radioactive for thousands of years and poses both environmental and security risks. Cesium and strontium can spread in the environment and affect human health by entering food and water systems.
In thorium-based reactors, while fission products like cesium-137 and strontium-90 are still present, the production of heavy long-lived elements like plutonium is much lower. Instead, isotopes like uranium-232 appear, which, although highly radioactive, decay faster and are less suitable for long-term accumulation.
The result is that thorium waste generally has lower long-term radiotoxicity, meaning it requires secure storage for shorter periods compared to uranium waste, even though strict handling is still necessary.
📊 Comparison Table: Uranium vs Thorium Reactors
| Aspect | Uranium Reactors | Thorium Reactors |
|---|---|---|
| Fuel Type | Direct fissile (U-235) | Fertile (Th-232 → U-233) |
| Process | Immediate fission | Multi-stage breeding + fission |
| Role of Half-Life | Less central to main fuel | Critical in fuel formation |
| Control | Requires external systems | More naturally regulated |
| Key By-products | Plutonium-239, Cs-137, Sr-90 | U-233 (with U-232), fewer actinides |
| Weaponization | Easier (Pu-239, enriched U-235) | Difficult (U-232 contamination) |
| Waste Duration | Very long-lived | Comparatively shorter-lived |
| Complexity | Lower | Higher (advanced engineering required) |
🧠 Final Understanding
Thorium and uranium both ultimately produce energy through nuclear fission, but they differ in how the process is structured. Uranium provides a direct and powerful reaction, with simpler fuel use but greater challenges in terms of weaponization risks and long-lived waste. It produces plutonium as a by-product, which can be used in nuclear weapons under certain conditions.
Thorium introduces an additional step—breeding governed by radioactive decay and half-life—which slows and regulates the formation of usable fuel. This makes the system inherently more controlled. At the same time, the presence of highly radioactive contaminants like uranium-232 makes thorium-derived materials difficult to weaponize, and its waste profile reduces long-term environmental burden.
For these reasons, thorium is considered a more controlled and strategically safer pathway for nuclear energy, especially when evaluated in terms of process behavior, resistance to weaponization, and waste management.